Blog: Digital Health and CNS Trials: A Promising Future Together
Much has been written and discussed about the impact that Digital Health is having on the evolution of clinical trials. While many agree that the COVID pandemic has greatly accelerated this process, others are split on the long-term impact Digital Health technologies project to ultimately have on patient outcomes. While it is still early days, experts have even wondered if Digital Health has failed to reach its potential, and subsequently will begin losing steam in the marketplace especially in light of the recent economic downturn. Most are pointing to anticipated spending cutbacks combined with the current healthcare systems struggles to evolve with innovation as key limitations to future patient impact and ROI.
Where does this leave clinical trials, known as the incubator of improved medical treatment? CNS trials, in particular, have been desperate for innovation as a result of the traditional methods utilized in drug discovery, research and treatment phases. A recent study suggested that CNS trials fail more than any other clinical trial investigation, for reasons that include a) strategy and planning, b) safety and toxicity, and c) lack of clinical efficacy. Most importantly, however CNS trials are failing due to the lack of reliable objective markers present in most neurodegenerative disorders. Ultimately this has forced CNS based pharmaceutical strategies to focus trials on the treatment of symptoms, rather than defined and measurable outcomes that impact disease detection and progression. Furthermore, without these identifiers, study outcomes are heavily reliant on the patient’s reported relief from symptoms as a determinant of efficacy.
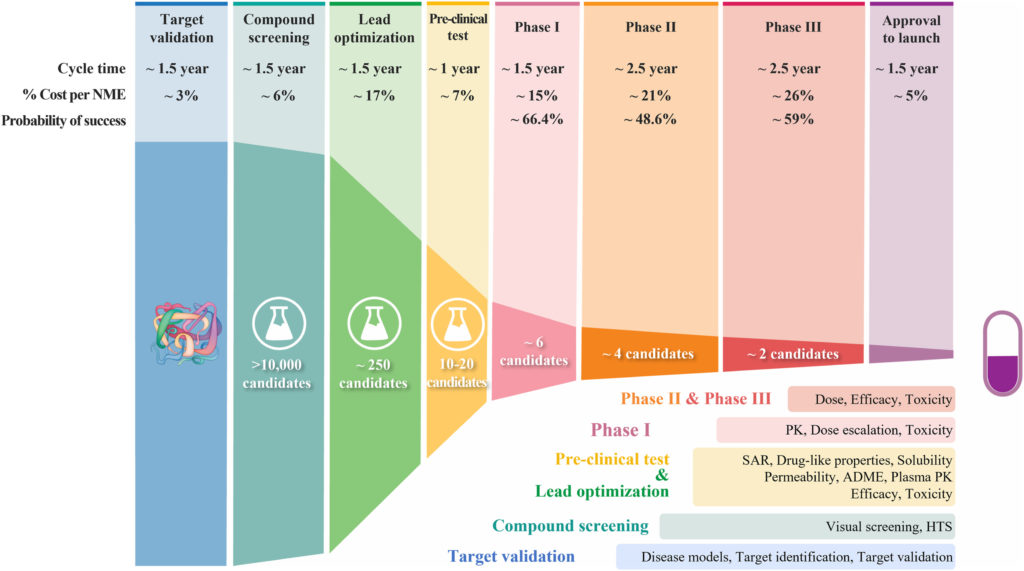
The drug development pipeline. Source: Sun Duxin et al., Why 90% of clinical drug development fails and how to improve it?,Acta Pharmaceutica Sinica B
In CNS trials, success rates are the worst in Parkinson’s Disease (PD), Alzheimer’s Disease, and Amyotrophic Lateral Sclerosis (ALS). While both AD and ALS drug trials have shown promise in animal models with respect to the identification and treatment of brain biomarkers, (known as beta-amyloid plaque that are thought to rapidly accelerate neuronal death, resulting in degeneration), clinically a strong relationship has emerged between the speed of disease progression and the long-term efficacy of many compounds, making them less effective over time. While there are many reasons why drugs fail to show clinical efficacy in later stage trials, scientists suspect that the reasons why CNS trials are failing 50% more than any other disease category are lost in the otherwise random onset nature of these disorders. In addition, of patients that are categorized as genetically predisposed often show no sign of cognitive impairment during trials, while those with correlated beta-amyloid plaque markers preclinically fail to show signs of disease progression during the trial period. This makes the process of capturing the ideal population at the right moment in time extremely challenging, and often leads to additional study burden as they linger beyond the trial period before ultimately failing.
Considering these inherent challenges, how can the promise of Digital Health technologies facilitate better outcomes for CNS trials and deliver better treatment options for patients?
Providing Real World Evidence (RWE)
Digital Health solutions often include wearable devices and other consumer technologies that are wirelessly enabled for long duration use. The benefits of deploying these systems in the context of trials is considerable, as patients can be monitored remotely and more effectively, and data collected during the normal waking hours of everyday life provides substantial RWE about how the patient is impacted by their condition and the subsequent changes that occur while under an experimental treatment. Regarding specific CNS endpoints, remote enabled solutions are among the most commonly utilized data collection methods in later stage clinical trials today, providing more options for meeting success criteria and advancing more compounds through the early stages.
Increased Accessibility
CNS trials are tainted by long study periods and even longer pre-market approvals from the FDA. Trials often exceed the initial study period due to a multitude of factors, however the most common issue is access to patients. Digital Health solutions inherently provide organizations with the ability to decentralize, allowing for greater geographic coverage that results in a larger population to draw subjects from. Even patients in remote areas are now accessible through emerging digital tools that can enhance participation but also improve retention.
Time and Cost Efficiencies
By establishing a larger study footprint and accessing a larger population, the time needed to complete trials can be reduced significantly. As a result, the operational burden for planning and executing trials inclusive of elaborate recruitment strategies is augmented considerably, allowing for less personnel, sites, and infrastructure. The patient burden is also reduced by requiring less paperwork and travel time. Both positively impact the bottom line, allowing for a more realistic strategy and planning phase.
SyncThink has been supporting CNS trials for many years, both as a CRO and as a preferred technology provider. We work closely with pharmaceutical companies and CRO’s looking to advance promising drug compounds in Phase I, II, or III trials through the use of our innovative biomarkers. These biomarkers combine with our wireless, mobile technology to accelerate trials, enable robust data collection at specified intervals, and deliver effective insights into drug performance on a host of neurological conditions.
If you’re interested in learning more about how we support CNS trials, contact us directly at info@syncthink.com